The Horizon 2020 FET-OPEN-funded project NanoTBTech is leading the way to new thermal bioimaging technologies.
Temperature measurements for biomedical technology have a two-pronged promise to fulfil, namely the detection and spatial mapping of temperature gradients for the better and earlier detection of diseases, and the real-time monitoring of hyperthermia treatments with the aim of making this a general non-invasive and innocuous treatment technique. For thermal bioimaging technology to tackle those two complex challenges, non-contact thermometry must be achieved with a sub-micrometre resolution; while providing high sensitivity thermal readout in a real-time mode. The NanoTBTech FET-OPEN project is leading the global effort to make these dreams come true.
The main pathways of the NanoTBTech approach are luminescent nanoparticles with high thermal sensitivity values, which are nontoxic, have long-circulating ‘stealth’, are functionalised, and are tumour-targeted. They are also non-invasive (as with NIR-to-NIR deep-tissue luminescence thermometry) and enable a real-time readout, both in vitro and in vivo.
Simple and compact prototypes coupling luminescent 2D time-resolved thermal imaging and optical microscopy imaging under NIR irradiation (or AC magnetic field) to monitor local hyperthermia in cells (CSIC), and to study in vivo time-gated and 2D hyperspectral magnetic- or optically-gated thermal transient thermometry in-depth tumour models (Biospace Lab/FIBIRYCIS), are currently being developed.
Accurate temperature readouts
Temperature plays a central role in the myriad of biochemical reactions regulating life. For instance, the intracellular temperature depends on cellular activity, including cell division, gene expression, enzymatic reactions, and pathological states. Cells have developed thermoregulation mechanisms to neutralise large external temperature changes and to maintain homeostasis of the body temperature upon exposure to hot or cold stimuli, an intriguing and not yet fully understood mechanism. NanoTBTech is tackling the issue of accurate temperature readouts, both in vivo and in vitro.
Although currently less mature than other, clinically battle-hardened imaging techniques such as magnetic resonance imaging (MRI) or positron emission tomography (PET), thermal bioimaging based on luminescence requires neither long scanning times nor post-processing analysis, providing a real-time readout far from the reach of all those competing techniques.
Achievements
During the last year (the project started in September 2018) NanoTBTech has reported several remarkable achievements, including:
- The design and synthesis of novel luminescent nanoparticles (Opt. Commun. 452, 342, 2019, Vinca) and heater-thermometer nanostructures (J. Phys. Chem. C 124, 8938, 2020, CNRS);
- The standardisation of light-to-heat conversion efficiency measurements (WPAS);
- The development of a general theoretical framework of single ion luminescent thermometers (UU); and
- The assessment of in vitro (CSIC/UAVR) and in vivo (FIBIRYCIS) temperature.
Optimum performance for thermal bioimaging
A general theoretical framework of single ion luminescent thermometers was developed, offering simple, user-friendly guidelines for both the choice of an appropriate emitter and the embedding host material for optimum temperature sensing (Adv. Theory Simul. 10.1002/adts.202000176 2020). The results show that the optimum performance (thermal response and thermal sensitivity) around temperature T0 is realised for an energy gap ∆E21 between thermally coupled levels (two and one) between 2kBT0 and 3.41kBT0 (kB is the Boltzmann constant). Analysis of the temperature-dependent excited state kinetics shows that host lattices in which ∆E21 can be bridged by one or two phonons are preferred over hosts in which higher-order phonon processes are required. Such a framework is relevant for both a fundamental understanding of luminescent thermometers but also the targeted design of novel and superior luminescent (nano)thermometers.
Accessing in vitro and in vivo temperatures
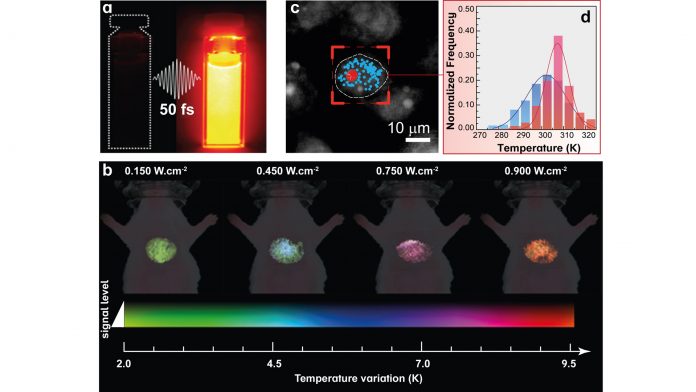
At in vivo level, the consortium reported a simple and straightforward analysis of emission bands of nanothermometers providing accurate and reliable thermal readouts (Adv. Funct. Mater. 2020, 10.1002/adfm.202002730). NanoTBTech researchers have found experimental evidence that the unique optical properties of living tissues may cause relevant spectral distortions in the recorded emission spectra from luminescent nanothermometers. The practical consequence of this is the traditional methods of thermal bioimaging may lead to large thermal readout errors. The research team have also explored several routes such as the fluorescence lifetime or emission intensity as thermal indicators, in both in vivo measurements and in silico simulations. These new approaches are exploited using also super-bright luminescent nanothermometers. One example, with great potential for in vivo measurements, uses Ag2S superdots fabricated by ultrafast photochemistry (Nat. Commun. 11, 2933, 2020) – see Fig. 1a. Thanks to their superior performance, it is now possible to measure the temperature of internal organs such as the liver (see Fig. 1b).
For in vitro temperature readouts, the NanoTBTech consortium reported the development of a unique tool for real-time bidimensional intracellular temperature mapping (Nano Lett. 20, 6466, 2020). The publication describes innovative thermal probes consisting of lanthanide-bearing polymeric micelles and their application in the temperature mapping of breast metastatic adenocarcinoma cells. An adapted fluorescence microscope was developed which permitted the real-time recording of the emission of the thermal probes, which is subsequently converted to the intracellular temperature. This singular approach allowed the observation of a higher temperature for the nucleolus (signed with red dots in Fig. 1c) relative to the rest of the cell (blue dots in Fig. 1c) and inhomogeneous intracellular temperature progressions (Fig. 1d, histograms obtained from the red and blue points indicated in panel c).
The challenge ahead: measuring temperature in the presence of an external magnetic field
The NanoTBTech consortium will proceed with in vitro and in vivo studies by quantifying the temperature gradients induced by a magnetic field. Under external alternating magnetic radiation, magnetic nanoparticles localised in a target organelle inside the cell or a living tissue induce a controlled and localised temperature increase, a decisive step forward towards the development of efficient novel magnetic hyperthermia cancer therapies. For in vitro, preliminary results demonstrated that temperature gradients are observed after exposure to the magnetic field. At present, experiments are underway to determine if these localised thermal gradients are enough to induce the cell apoptosis, achieving consequently the desirable therapeutic effect.
NanoTBTech’s partners
CICECO – Aveiro Institute of Materials, Universidade de Aveiro (UAVR – Portugal) is co-ordinating the project. Eight other partners participate in NanoTBTech:
- Fundacion para la Investigacion Biomedica del Hospital Universitario Ramon Y Cajal (FIBIRYCIS – Spain);
- Centre National de la Recherche Scientifique (CNRS – France);
- Agencia Estatal Consejo Superior de Investigaciones Cientificas (CSIC – Spain);
- Institut Za Nuklearne Nauke Vinca (Vinca – Serbia);
- Instytut Niskich Temperatur I Badan Strukturalnych Im. Wlodzimierza Trzebiatowskiego Polskiej Akademii Nauk (WPAS – Poland);
- Universiteit Utrecht (UU – The Netherlands);
- Nanoimmunotech Sl (NIT – Spain); and
- Biospace Lab SA (Biospace Lab – France).
This project has received funding from the European Union’s Horizon 2020 FET Open programme under grant agreement No 801305.
Luís D. Carlos
Full Professor
University of Aveiro
CICECO – Aveiro Institute of Materials (Phantom-g)
+351 234 370946
lcarlos@ua.pt
Tweet @nanotbtech
www.nanotbtech.eu