The Karlsruhe Institute of Technology discuss their method of promoting battery safety, which holds the potential to revolutionise the transport industry.
Battery calorimeters are essential to ensuring the safety of batteries. By monitoring the thermal runaway, technicians can prevent ignition or even explosion of the cell which can release toxic gases.
In the last nine years, the Karlsruhe Institute of Technology have established battery calorimetry at the IAM-AWP Calorimeter Center as a powerful and versatile electrochemical-thermal characterisation technique, which allows advancements for the thermal management, the lifetime and the safety of batteries.
The IAM-AWP Calorimeter Center operates Europe’s largest battery calorimeter laboratory and provides six Accelerating Rate Calorimeters (ARCs) of different sizes – from coin to large pouch or prismatic automotive format – which allow the evaluation of thermodynamic, thermal and safety data for Lithium-ion cells on material, cell and pack level under quasiadiabatic and isoperibolic environments for both normal and abuse conditions (thermal, electrical, mechanical).
Ageing studies
For ageing studies, the cell is inserted into the calorimeter chamber and coupled to a battery cycler. Isoperibolic or quasiadiabatic ambient conditions are adjusted by heaters and thermocouples, that are found in lid, bottom, and sidewalls of the chamber. Isoperibolic means, that the temperature of the chamber is kept constant and the temperature change at the surface of the cell is measured. In this case, the cell temperature reaches its initial temperature again after each cycle.
In the quasiadiabatic mode the heaters follow immediately any change of the cell temperature preventing the heat transfer to the chamber. This simulates ambient conditions for a cell in a pack, where the densely packed neighbouring cells, prevent, or at least greatly reduce, the heat release to the environment. Such conditions lead to a continuous increase of the cell temperature with every cycle.
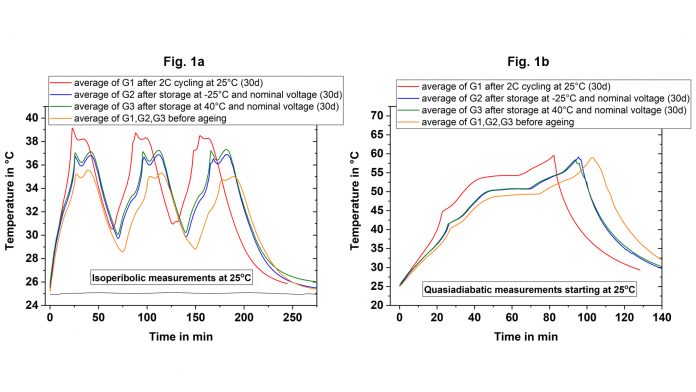
Using these two conditions, an ageing study was carried out. First, fresh cylindrical 18650 cells were stored at different temperatures in temperature chambers and then characterised at fixed time intervals in order to investigate the influence of this storage (calendar ageing) on cell performance. Secondly, cells were aged at a constant charging/discharging rate in temperature chambers (cyclic aging) at room temperature. Fig. 1 compares the temperature curves measured in the ARC between the cells in new condition and after cyclic (G1) or calendaric (G2, G3) ageing under isoperibolic conditions (Fig. 1a) or adiabatic conditions (Fig. 1b).
Under both conditions, cyclic ageing leads to the largest changes, which manifests under isoperibolic conditions as an increase in the maximum temperature by 4°C and under adiabatic conditions by reaching the maximum permitted operating temperature of 60°C earlier. Calendar ageing also leads to the same effects, however, regardless of the storage temperature, the increase in the maximum temperature is only 1.5°C. In addition, cyclic ageing also changes the curve shape, which is a further indicator for the ageing status or history. Thus, recording of the temperature profile can be used as a “fingerprint” for the state-of-health (SOH) and as fast and reliable method for the characterisation of ageing processes.
Entropy measurement and electrochemical impedance spectroscopy
Once the cells have reached their termination criterion, the reversible and the irreversible heat are measured using entropy measurement and electrochemical impedance spectroscopy (EIS). These data can be used for the parameterisation of impedance-based electrical equivalent circuit models (EECMs) and ageing models. In addition, some cells are analysed post-mortem, i.e. disassembled in an inert gas atmosphere and the changes in the chemical composition, the morphology and the structure of the electrodes are determined. Moreover, the influence of ageing and operating conditions on cell safety can be addressed in the ARCs. This means that the change in the hazard potential of the cells with increasing degree of ageing can be quantitatively determined.
To summarise, battery calorimetry is now well established and is increasingly becoming more requested by our European research partners from both industry and academia, because quantitative and system relevant data for temperature, heat and pressure development of materials and cells are provided as a fast feedback for the cell developers and as input data for modelling and simulation.
There are still numerous challenges to address and we hope that our Calorimeter Center will help the European industry to make further progress in the battery field, which is urgently needed to reach a low-carbon future, to foster European leadership, and to create new jobs, which are the main objectives of the European Battery Alliance.