Next generation extracellular matrix biomaterials promote healing – Is human tissue regeneration within reach?
Unlike less-developed species such as the axolotl and zebra fish, the adult human has limited ability to regenerate tissue following injury.1, 2 Select tissues such as the liver, bone marrow, and the inner lining of the intestinal tract retain regenerative potential in humans, but most other tissues respond to injury (heal) via well the understood processes of hemostasis, inflammation, and deposition of scar tissue. Of note, the developing human foetus and neonate have a more robust ability to regenerate tissue/organs than adults; a healing characteristic rapidly lost following birth. This fact is evidence for the genetically endowed potential for regeneration, but the signals required to activate and realise this potential in adults are unknown.
Why did the evolution of Homo sapiens include the loss of regenerative capability and replacement by scar tissue formation?
The most common answer to this question is that there was an evolutionary survival advantage for this trade off. Risk of exsanguination and infection following traumatic injury drove the selection of a swift, albeit imperfect healing process (i.e. haemostasis and scarring) over regeneration. This exchange for survival comes with the cost of increased morbidity and decreased tissue functionality, both of which are dependent upon the location and extent of injury. A clinical example of the pros and cons of this evolutionary selection can be found in patients with traumatic injury in which massive amounts of extremity muscle tissue are destroyed or lost (i.e., volumetric muscle loss). The ability to halt the haemorrhage by blood clot formation and overcome associated wound infection avoids loss of life, but the resulting replacement of skeletal muscle by scar tissue markedly compromises subsequent limb function.
Are there therapeutic strategies that compensate for loss of regenerative ability?
Many strategies for functional tissue replacement exist including cell-based therapies, 3D printing, and tissue/organ culture, however successful clinical translation of these approaches has been very limited. An alternative strategy involves the resurrection or reactivation of at least some of the inherent regenerative capacity that exists in the human genome. Can therapeutic approaches be developed that both retain immediate life-preserving processes and simultaneously activate the molecular signals required to promote and facilitate the otherwise silent genetically-driven pathways of tissue regeneration?
To answer these questions, we turn to the immune system. Once thought to have the singular function of protection against pathogens and tissue injury, studies within the past two decades have identified the critical role the immune system plays in normal tissue development and tissue regeneration. Immune system function in higher mammals, including both the innate and adaptive arms, is tightly linked to wound healing and inherent regenerative potential and must be spatially and temporally regulated for any true regeneration to occur. Without such regulation, scarring results, limiting regenerative capacity.
There is some evidence that evolution of the adaptive immune system is inversely correlated with regenerative capacity.3 Though the true link between these two evolved and devolved traits is not well understood, the shared trafficking mechanisms between immune cells and stem/progenitor cells that contribute to regeneration may be a contributing factor and could in part, explain the inverse relationship between immune competence and robust regeneration.4
While the addition of a highly specialised, adaptive immune system brings improved host defence against pathogens like viruses, bacteria, etc., some speculate that this defence function may come with a trade-off of full regenerative potential. In foetal development for example, the balance between immune competence and regeneration favours regeneration, a function largely lost in later life. Of note, this trade-off may provide for greater protection also against cancer development and metastasis, which involves many of the same mechanisms as immune trafficking required for robust regeneration.
Therapeutic approaches to stimulate regeneration should be cognisant of these trade-offs; nonetheless, important clues continue to be unveiled as our understanding of concomitant development of specialised immune function and regenerative capacity expands.
Regenerative Signals within ECM
It is recognised that molecular signals originating in the extracellular matrix (ECM) can modulate the seemingly mutually exclusive processes of immunity and tissue regeneration, although the specific bioactive molecules have yet to be identified.
The ECM is a rich source of signalling molecules for both the resident cells and cells that transit through the ECM in all tissues. These signals maintain a cell-friendly homeostatic microenvironment during states of health and mitigate the proinflammatory response and facilitate functional tissue reconstruction following injury. Isolation of the ECM from tissues such as dermis, small intestine, and urinary bladder, among others, by removal of cells (decellularisation) allows for the creation of ECM-based biomaterials. Such ECM-based materials are currently available as surgical meshes for ventral hernia repair, musculotendinous repair,5-9 and dura mater replacement,10, 11 among other therapeutic applications,12-15 and as powders for topical wound care.16, 17 These embedded signalling molecules are released during the process of ECM degradation in vivo. These signalling molecules favourably modulate the innate immune response and provide a microenvironment that influences cellular differentiation and spatial organisation.18-22 The clinical outcome includes partial restoration of site appropriate normal tissue structure and function.
Using the principles of Mother Nature to restore healthy tissue
The concept of the microenvironment, and specifically the ECM, regulating cell and tissue morphogenesis is not new. Bissell and Sonnenschein have studied these concepts primarily in the context of cancer, focused upon the direct cell-matrix bi-directional crosstalk without mention of the indirect and paracrine influence of the immune system. Sonnenschein has referred to the temporal and spatial effects of the microenvironmnet upon cell behaviour as the tissue organisation field theory’ (TOFT). 23, 25, 26, 27
One example of a clinical application in which the ECM, when used as a biomaterial, can redirect dysregulated cell-signalling mechanisms involves its therapeutic use for the treatment of oesophageal adenocarcinoma.15 Following full circumferential mucosal resection of the cancerous mucosa, an ECM bioscaffold was implanted in five patients. Patients achieved complete regeneration of a near-normal oesophageal mucosa and remained cancer free after a follow-up of more than five years. The use of this acellular regenerative medicine approach avoided the highly morbid procedure of esophagectomy, stricture (i.e. scar tissue) formation, and cancer progression. The restoration of normal or near normal tissue is an example of modulating the default tissue repair pathways toward activation of endogenous constructive and functional repair pathways by utilisation of Mother Nature’s signals embedded within the ECM. This approach is a viable alternative to tissue engineering/regenerative medicine approaches that involve cell-based therapy or exogenous tissue/organ creation with subsequent surgical implantation.
A second clinical example of using naturally occurring signals to promote functional tissue restoration involves the use of ECM bioscaffolds for skeletal muscle repair. 13 patients with volumetric muscle loss had exhausted all attempts to replace missing muscle tissue with conventional therapies.14 Following implantation of a surgical mesh composed of ECM, the average patient showed a 37% increase in strength, 38% restoration of muscle mass, and greater the 250% improvement in ability to perform muscle functional activities. This cohort study followed extensive preclinical work in which ECM signalling molecules were shown to mitigate the proinflammatory immune response and promote the proliferation and differentiation of muscle progenitor cells.22, 28, 29
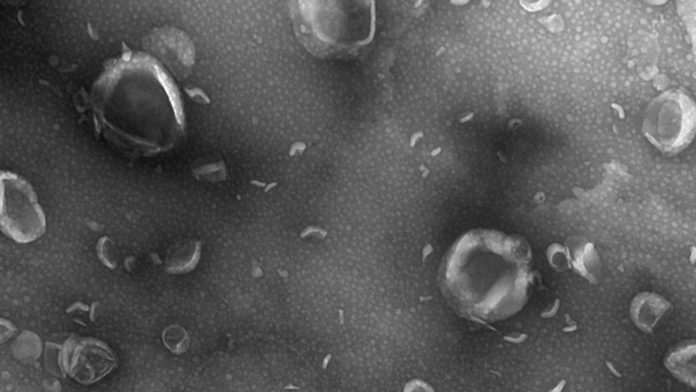
The next generation of ECM-based therapeutics will undoubtedly involve the identification and use of various formulations of ECM, such as hydrogels that can be injected by minimally invasive methods, and the use of specific components of the ECM that are shown to possess immunomodulatory properties. An example of the latter is the recent identification of matrix bound nanovesicle’s (MBV).30 These nano-sized, lipid bound vesicles are firmly embedded within the structural components of the ECM and are a distinct subset of extracellular vesicles from the more commonly described exosomes found in body fluids.31 MBV have potent immunomodulatory properties and are only released from their ECM microenvironment when the matrix is degraded, as occurs following tissue injury or during the degradation of an ECM biomaterial. The size and abundance of MBV provide great flexibility for therapeutic delivery and expansion of the use of naturally occurring signalling molecules to restore healthy tissue.32
Advancements in surgical technique, 3D printing of tissues and organs, bioreactor technology, stem cell biology and related disciplines will clearly have a positive impact upon the clinical translation of regenerative medicine. It is just as evident that our understanding and ability to influence naturally occurring pathways of tissue development and regeneration through the use of ECM-based signals and related technologies will have a significant impact upon future therapeutic approaches.
Intellectual property related to the hydrogel form of extracellular matrix and matrix bound nanovesicles has been licensed to ECM Therapeutics, Inc., a company in which SFB, and JLD have a vested interest and hold officer positions.
References
- W. Seifert, K. Muneoka, ‘The blastema and epimorphic regeneration in mammals’, Dev Biol 433(2) (2018) 190-199
- A. Eming, P. Martin, M. Tomic-Canic, ‘Wound repair and regeneration: mechanisms, signaling, and translation’, Sci Transl Med 6(265) (2014) 265sr6
- W. Godwin, A.R. Pinto, N.A. Rosenthal, ‘Chasing the recipe for a pro-regenerative immune system’, Semin Cell Dev Biol 61 (2017) 71-79
- Wang, H. Knaut, ‘Chemokine signaling in development and disease’, Development 141(22) (2014) 4199-205
- A. Kissane, K.M. Itani, ‘A decade of ventral incisional hernia repairs with biologic acellular dermal matrix: what have we learned?’, Plast Reconstr Surg 130(5 Suppl 2) (2012) 194S-202S
- Badylak, S. Arnoczky, P. Plouhar, R. Haut, V. Mendenhall, R. Clarke, C. Horvath, ‘Naturally occurring extracellular matrix as a scaffold for musculoskeletal repair’, Clin Orthop Relat Res (367 Suppl) (1999) S333-43
- J. Turner, J.S. Badylak, D.J. Weber, S.F. Badylak, ‘Biologic scaffold remodeling in a dog model of complex musculoskeletal injury’, J Surg Res 176(2) (2012) 490-502
- J. Turner, S.F. Badylak, ‘Biologic scaffolds for musculotendinous tissue repair’, Eur Cell Mater 25 (2013) 130-43
- J. Turner, A.J. Yates, Jr., D.J. Weber, I.R. Qureshi, D.B. Stolz, T.W. Gilbert, S.F. Badylak, ‘Xenogeneic extracellular matrix as an inductive scaffold for regeneration of a functioning musculotendinous junction’ Tissue Eng Part A 16(11) (2010) 3309-17
- A. Cobb, S.F. Badylak, W. Janas, F.A. Boop, ‘Histology after dural grafting with small intestinal submucosa’, Surg Neurol 46(4) (1996) 389-93; discussion 393-4
- A. Cobb, S.F. Badylak, W. Janas, A. Simmons-Byrd, F.A. Boop, ‘Porcine small intestinal submucosa as a dural substitute’, Surg Neurol 51(1) (1999) 99-104
- E. Mewhort, J.D. Turnbull, H.C. Meijndert, J.M. Ngu, P.W. Fedak, ‘Epicardial infarct repair with basic fibroblast growth factor-enhanced CorMatrix-ECM biomaterial attenuates postischemic cardiac remodeling’, The Journal of thoracic and cardiovascular surgery 147(5) (2014) 1650-9
- A. Derwin, S.F. Badylak, S.P. Steinmann, J.P. Iannotti, ‘Extracellular matrix scaffold devices for rotator cuff repair’, J Shoulder Elbow Surg 19(3) (2010) 467-76
- Dziki, S. Badylak, M. Yabroudi, B. Sicari, F. Ambrosio, K. Stearns, N. Turner, A. Wyse, M.L. Boninger, E.H.P. Brown, J.P. Rubin, ‘An acellular biologic scaffold treatment for volumetric muscle loss results of a 13-patient cohort study’, NPJ Regen Med 1 (2016) 16008
- F. Badylak, T. Hoppo, A. Nieponice, T.W. Gilbert, J.M. Davison, B.A. Jobe, ‘Esophageal preservation in five male patients after endoscopic inner-layer circumferential resection in the setting of superficial cancer: a regenerative medicine approach with a biologic scaffold’, Tissue Eng Part A 17(11-12) (2011) 1643-50
- D. Prevel, B.L. Eppley, D.J. Summerlin, R. Sidner, J.R. Jackson, M. McCarty, S.F. Badylak, ‘Small intestinal submucosa: utilization as a wound dressing in full-thickness rodent wounds’, Ann Plast Surg 35(4) (1995) 381-8
- J. Turner, S.F. Badylak, ‘The Use of Biologic Scaffolds in the Treatment of Chronic Nonhealing Wounds’, Adv Wound Care (New Rochelle) 4(8) (2015) 490-500
- P. Brennan, J. Reing, D. Chew, J.M. Myers-Irvin, E.J. Young, S.F. Badylak, ‘Antibacterial activity within degradation products of biological scaffolds composed of extracellular matrix’, Tissue Eng 12(10) (2006) 2949-55
- P. Brennan, X.H. Tang, A.M. Stewart-Akers, L.J. Gudas, S.F. Badylak, ‘Chemoattractant activity of degradation products of fetal and adult skin extracellular matrix for keratinocyte progenitor cells’, J Tissue Eng Regen Med 2(8) (2008) 491-8
- T. Saldin, S. Patel, L. Zhang, L. Huleihel, G.S. Hussey, D.G. Nascari, L.M. Quijano, X. Li, D. Raghu, A.K. Bajwa, N.G. Smith, C.C. Chung, A.N. Omstead, J.E. Kosovec, B.A. Jobe, N.J. Turner, A.H. Zaidi, S.F. Badylak, ‘Extracellular Matrix Degradation Products Downregulate Neoplastic Esophageal Cell Phenotype’, Tissue Eng Part A 25(5-6) (2019) 487-498
- Tottey, M. Corselli, E.M. Jeffries, R. Londono, B. Peault, S.F. Badylak, ‘Extracellular matrix degradation products and low-oxygen conditions enhance the regenerative potential of perivascular stem cells’, Tissue Eng Part A 17(1-2) (2011) 37-44
- M. Sicari, J.L. Dziki, B.F. Siu, C.J. Medberry, C.L. Dearth, S.F. Badylak, ‘The promotion of a constructive macrophage phenotype by solubilized extracellular matrix’, Biomaterials 35(30) (2014) 8605-12
- J. Bissell, M.H. Barcellos-Hoff, ‘The influence of extracellular matrix on gene expression: is structure the message?’, J Cell Sci Suppl 8 (1987) 327-43
- M. Ghajar, H. Peinado, H. Mori, I.R. Matei, K.J. Evason, H. Brazier, D. Almeida, A. Koller, K.A. Hajjar, D.Y. Stainier, E.I. Chen, D. Lyden, M.J. Bissell, ‘The perivascular niche regulates breast tumour dormancy’, Nat Cell Biol 15(7) (2013) 807-17
- D. Roskelley, M.J. Bissell, ‘Dynamic reciprocity revisited: a continuous, bidirectional flow of information between cells and the extracellular matrix regulates mammary epithelial cell function’, Biochem Cell Biol 73(7-8) (1995) 391-7
- M. Markey, M.A. Coombs, C. Sonnenschein, A.M. Soto, ‘Mammalian development in a changing environment: exposure to endocrine disruptors reveals the developmental plasticity of steroid-hormone target organs’, Evol Dev 5(1) (2003) 67-75
- M. Soto, C. Sonnenschein, ‘Emergentism as a default: cancer as a problem of tissue organization’, J Biosci 30(1) (2005) 103-18
- L. Dziki, R.M. Giglio, B.M. Sicari, D.S. Wang, R.M. Gandhi, R. Londono, C.L. Dearth, S.F. Badylak, ‘The Effect of Mechanical Loading Upon Extracellular Matrix Bioscaffold-Mediated Skeletal Muscle Remodelling’, Tissue Eng Part A (2017)
- L. Dziki, B.M. Sicari, M.T. Wolf, M.C. Cramer, S.F. Badylak, ‘Immunomodulation and Mobilization of Progenitor Cells by Extracellular Matrix Bioscaffolds for Volumetric Muscle Loss Treatment’, Tissue Eng Part A (2016)
- Huleihel, G.S. Hussey, J.D. Naranjo, L. Zhang, J.L. Dziki, N.J. Turner, D.B. Stolz, S.F. Badylak, ‘Matrix-bound nanovesicles within ECM bioscaffolds’, Sci Adv 2(6) (2016) e1600502
- S. Hussey, C. Pineda Molina, M.C. Cramer, Y.Y. Tyurina, V.A. Tyurin, Y.C. Lee, S.O. El-Mossier, M.H. Murdock, P.S. Timashev, V.E. Kagan, S.F. Badylak, ‘Lipidomics and RNA sequencing reveal a novel subpopulation of nanovesicle within extracellular matrix biomaterials’, Sci Adv 6(12) (2020) eaay4361
- S. Hussey, J.L. Dziki, Y.C. Lee, J.G. Bartolacci, M. Behun, H.R. Turnquist, S.F. Badylak, ‘Matrix bound nanovesicle-associated IL-33 activates a pro-remodelling macrophage phenotype via a non-canonical, ST2-independent pathway,’ J Immunol Regen Med 3 (2019) 26-35
Professor Stephen F Badylak
McGowan Institute for Regenerative Medicine
University of Pittsburgh
ECM Therapeutics
118 Marshall Drive
Warrendale, PA 15086
+1 412 624-5308
badysx@UPMC.EDU
Tweet @McGowanRM
https://mirm-pitt.net
www.ecmtherapeutics.com
Co-author
Jenna Dziki
Research Assistant Professor
McGowan Institute for Regenerative Medicine
University of Pittsburgh
Please note, this article will also appear in the second edition of our new quarterly publication.